Paleobotany
The Evolution of Plants
From flowerless forests dominated by giant conifers to grasslands covering 40% of the Earth. The history of plants throughout the Mesozoic and Cenozoic is as dramatic as that of the dinosaurs, and the two are deeply intertwined.
Forests without flowers
The plant world of the dinosaurs was radically different from ours. No grasses, no flowers, no fruits.
During the Triassic and Jurassic, the forest canopy was dominated by giant conifers of the Araucariaceae and Cheirolepidiaceae families, reaching 30 to 60 meters. Below, cycads, bennettitales, and ginkgoales formed the mid-story, and the ground was covered by ferns, mosses, and horsetails. There were no grasses, flowers, or fruits. This landscape persisted for over 100 million years.
Sauropod diets depended on these plants. Carbon isotope analyses from tooth enamel (Morrison Formation, USA) indicate that Brachiosaurus reached the araucaria canopy, while Diplodocus grazed on ground-level ferns. Laboratory tests (Hummel et al., 2008) showed that Araucaria foliage and horsetails produce energy comparable to modern fodder, making the gigantism of these animals viable.
Notable fossil sites
Petrified Forest, Arizona (~225 Ma): silicified trunks of Late Triassic conifers.
Cerro Cuadrado, Patagonia (~160 Ma): Araucaria mirabilis cones with cellular preservation and intact embryos.
Skåne, Sweden: Jurassic ferns with preserved cell nuclei and chromosomes (Bomfleur et al., 2014).
Jurassic forest vs. modern
The "flowers" before flowers: Bennettitales
An extinct order that developed flower-like structures completely independently from angiosperms. A remarkable case of convergent evolution.
Bennettitales were an order of gymnosperm plants that lived for nearly 170 million years. Externally resembling modern cycads, with robust trunks and pinnate leaves, they concealed a crucial difference: their reproductive organs. Some species, such as Williamsonia and Cycadeoidea, developed flower-like structures with petaloid bracts and male and female organs united in the same reproductive unit.
The decline of Bennettitales is closely tied to the rise of angiosperms. As flowering plants diversified from ~100 Ma onward, they conquered the ecological niches previously occupied by Bennettitales. With faster growth rates and mutualistic relationships with pollinators, angiosperms held a decisive competitive advantage. Bennettitales disappeared at the K-Pg boundary (66 Ma), possibly already in severe decline before the Chicxulub impact.
Convergent evolution
The former "Anthophyte Hypothesis" proposed that Bennettitales were direct ancestors of flowering plants. Modern phylogenetic analyses refuted this idea: Bennettitales are closer to cycads and ginkgos, not to angiosperms. The floral resemblance is purely convergent, which makes the group even more fascinating.
Two families
Williamsoniaceae
Branching trunks, exposed "flowers" with petaloid bracts. Visually similar to angiosperm flowers.
Cycadeoidaceae
Compact barrel-shaped trunks. Reproductive structures embedded in the surface, often closed (cleistogamy).
The "abominable mystery": the angiosperm revolution
Charles Darwin, in an 1879 letter to Joseph Hooker, called the explosive emergence of flowering plants an "abominable mystery." In 30 million years, they went from marginal to dominant.
"The rapid development as far as we can judge of all the higher plants within recent geological times is an abominable mystery."
Charles Darwin, letter to Joseph Hooker, July 22, 1879
The earliest angiosperm fossil records include pollen grains from ~136 million years ago found in Israel, the aquatic plant Montsechia vidalii (~130 Ma, Spain), and Archaefructus sinensis (~125 Ma, China). In just 30 million years, they went from marginal elements to dominants in terrestrial ecosystems, one of the fastest ecological transitions in history.
The impact on fauna was profound. The diversification of ornithischian dinosaurs in the Late Cretaceous coincides with angiosperm dominance. Hadrosaurs ("duck-billed" dinosaurs) developed complex dental batteries, with hundreds of compacted teeth forming grinding surfaces to process the tough, silica-rich leaves of the new plants.
Earliest fossils
Angiosperm pollen grains in Israel and Switzerland
Montsechia vidalii, aquatic plant from Spain (Gomez et al., 2015)
Archaefructus sinensis, Yixian Formation, China (Sun et al., 2002)
Decisive adaptations
Xylem vessels: more efficient water transport than conifer tracheids
Broad leaves: higher vein density, maximizing photosynthesis
Insect pollination: precise reproduction and accelerated speciation
Fruits: seed dispersal by animals, colonizing new habitats
Double fertilization: produces endosperm only when fertilization occurs, conserving energy
Flowers and pollinators: coevolution
The relationship between flowers and pollinating insects is one of the most successful partnerships in evolution. And it began in the world of dinosaurs.
The earliest evidence of insect pollination dates back to ~99 million years ago, preserved in Burmese amber (Myanmar). Fossils of thrips (Gymnopollisthrips minor) carrying pollen grains and beetles (Cretoparacucujus cycadophilus) associated with cycad pollen demonstrate that entomophilous pollination arose even before the great angiosperm diversification. Long-proboscid flies also appear in this period.
Insect pollination dramatically accelerated angiosperm diversification. By enabling reproductive isolation and rapid speciation, each flower-pollinator pair could diverge independently. Molecular clock studies (Magallón et al., 2015) show that the major angiosperm lineages (rosids, asterids) diversified between 115 and 90 Ma, coinciding with pollinator diversification.
Progression of pollination syndromes
Beetles
Oldest; visiting gymnosperm cones
Flies
Long proboscises in the mid-Cretaceous (~99 Ma)
Bees
Melittosphex burmensis (~100 Ma), the oldest known bee
Butterflies and moths
Moths with proboscis since ~130 Ma; butterflies in the Paleogene
Key evidence: Burmese amber (~99 Ma)
Multiple insect groups preserved carrying pollen: thrips, beetles, flies. Myanmar amber is Cenomanian in age and also contains one of the oldest flowers preserved in resin, Micropetasos burmensis, with structures indicating insect pollination.
Dinosaurs and angiosperms: coevolution or coincidence?
Flowering plants exploded through the Cretaceous, rising from 0 to 85% of the typical flora in about 65 million years. Dinosaurs reached their own diversity peak in the same window. Mike Benton, in a 2024 review of the "dinosaur boom in the Cretaceous," examined whether the two phenomena are causally linked or merely concurrent.
Angiosperms and gymnosperms are sister groups: the phylogenomic evidence requires that the angiosperm stem lineage originated at the same time as gymnosperms, at the latest by the Late Triassic (Li et al. 2019). But the earliest widely accepted fossils, characteristic pollen of the Valanginian-Hauterivian (140 to 130 Ma), only appear at the start of the Cretaceous. Macroscopic angiosperm remains, including fossil flowers, leaves, seeds, and other organs, show up from the Barremian-Albian (129 to 100 Ma) onward.
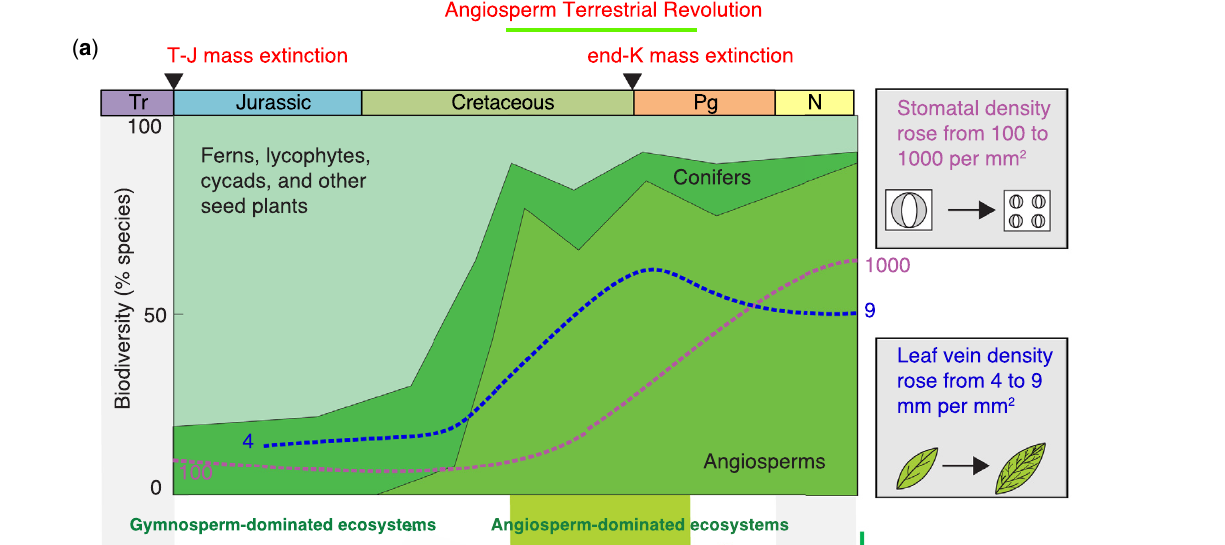
Why did angiosperms succeed so fast? Benton lists unique traits that set them apart: double fertilization (which only builds a seed once fertilization is confirmed), flower and coevolution with insect pollinators and, decisively, two Late Cretaceous innovations in leaf physiology. Stomatal density rose from about 100 to 1,000 pores per mm², and leaf vein density rose from roughly 4 to 9 mm of vein per mm² of leaf. Both are adaptations for faster uptake of CO₂ and faster transport of water and nutrients. Together, they made angiosperms photosynthetically more efficient than the gymnosperms they were replacing.
The Angiosperm Terrestrial Revolution (ATR)
Benton and colleagues (2022) named the ecological expansion of flowering plants the Angiosperm Terrestrial Revolution and dated it to 100 to 50 million years ago, split into two phases. The Cretaceous phase (100 to 66 Ma) is what matters for dinosaur evolution: angiosperms went from a marginal presence to dominance of the typical flora in roughly 34 million years. The second phase, in the Paleocene after the asteroid impact, marked the origin of tropical rainforests. The crossing of the 50% line, from gymnosperm to angiosperm dominance, happens near the Cenomanian-Turonian boundary, around 94 Ma.
Did dinosaurs drive the angiosperm revolution?
The classic hypothesis is Robert Bakker's (1978, 1986): sauropods were high browsers that stripped leaves from the canopy and did not feed close to the ground. At the start of the Cretaceous, many sauropod clades declined and the new dominant herbivores, ornithopods and nodosaurid ankylosaurs, fed at ground level. They would have grazed slow-growing gymnosperm seedlings, opening space for faster-growing angiosperms. In Bakker's scenario, angiosperm success is a consequence of a dinosaur changing of the guard.
Later studies questioned the chronology. Butler and colleagues (2009, 2010) found little repeatable evidence of coevolution between dinosaur groups and angiosperms when they tested temporal and spatial coincidences of occurrence across the Cretaceous record. And sauropods did not disappear across the Jurassic-Cretaceous boundary: they still made up 25 to 50% of herbivorous diversity through the Early Cretaceous (Barrett & Willis 2001). The "major sauropod exit" turned out to be gradual and regional, not abrupt and global.
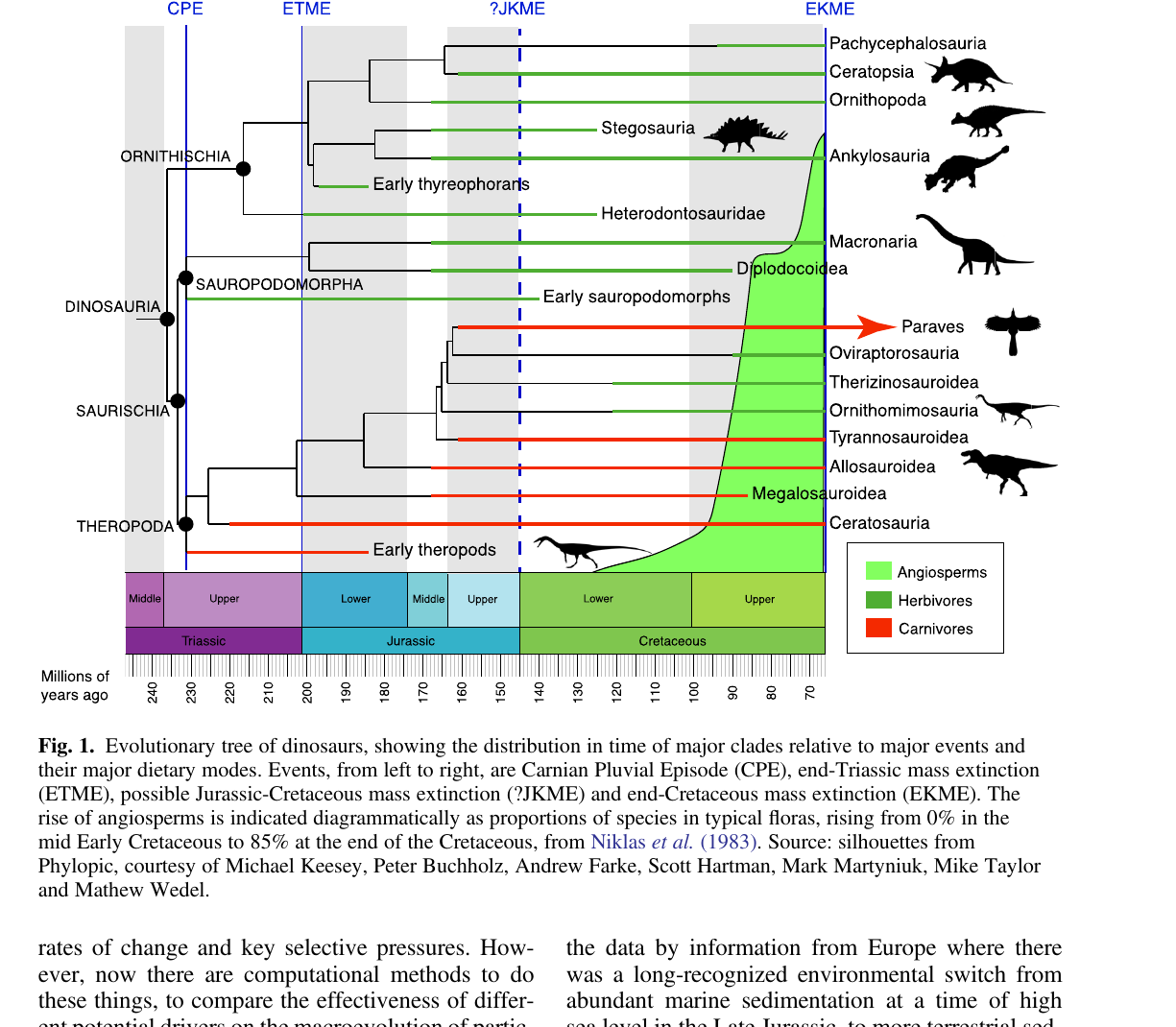
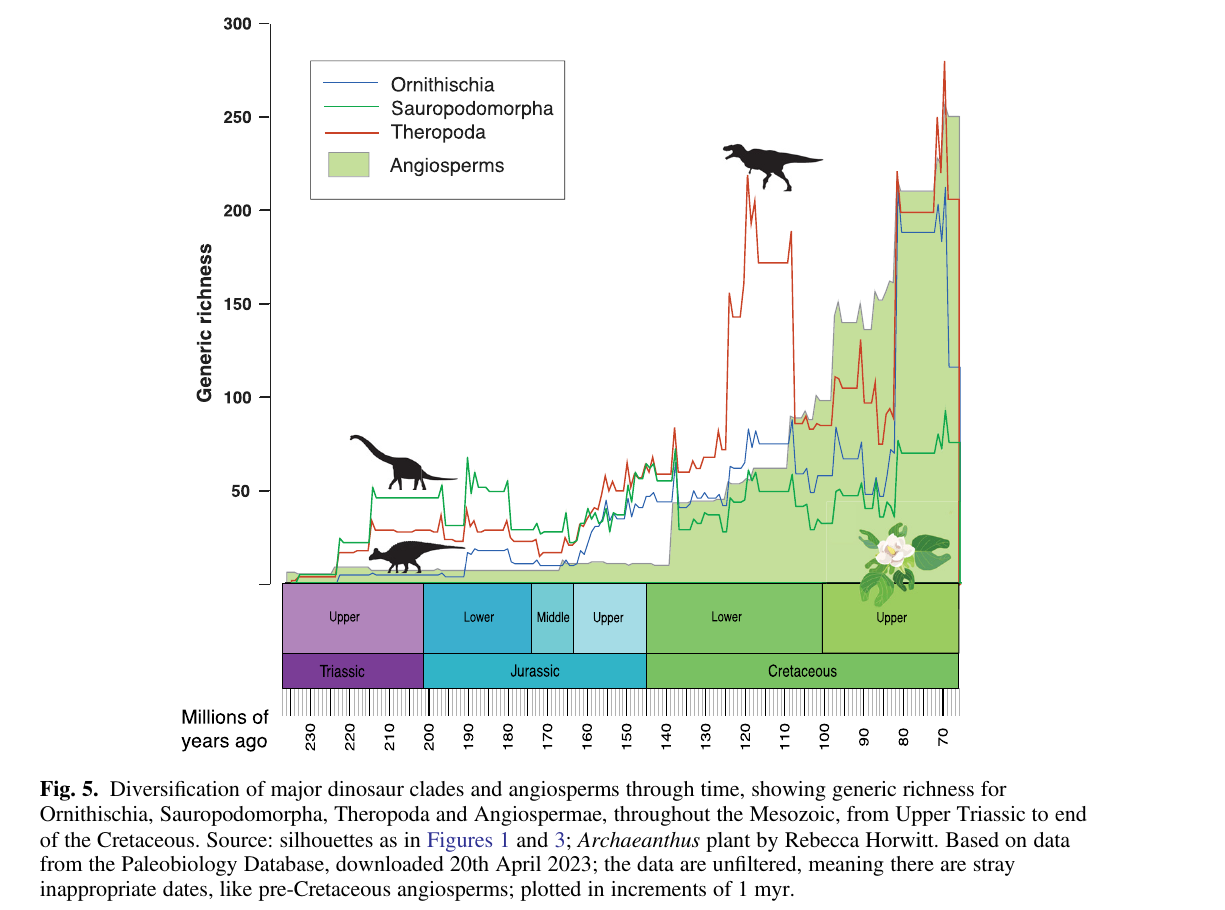
Benton's review compares diversification curves of the three major dinosaur clades against the angiosperm curve, using generic richness from the Paleobiology Database. The pattern is suggestive but not tight.
Two things stand out in Figure 5. Sauropodomorph richness is essentially flat at 40 to 80 genera through the entire Cretaceous. If angiosperms had been reshaping the herbivore niche, sauropodomorph diversity should move, up or down, in step with the floral turnover. It does not. Ornithischians, the clade Bakker put at the center of his story, only explode in diversity at the very end of the Cretaceous, long after the angiosperm rise. The theropod curve correlates with the angiosperm curve better than either herbivore curve does, which is hard to explain through direct herbivory pressure.
Direct evidence: what did dinosaurs actually eat?
Although the global curves do not show tight coevolution, specific gut and dung contents leave no doubt that dinosaurs ate angiosperms when they were available. Benton highlights three of the clearest cases:
Ankylosaur Minmi
Early Cretaceous (Aptian, around 112 Ma) of Australia. Preserved gut contents were composed entirely of angiosperm fruits (Molnar & Clifford 2000).
Hadrosaur coprolites
Campanian of Montana. Angiosperm-specific molecular biomarkers (Chin & Brassell 1994), alongside evidence that the animals also ate rotting conifer wood (Chin 2007). Mixed diet.
Titanosaur coprolites
Latest Maastrichtian of India. Contained angiosperm-specific parasitic fungi, grass phytoliths, and phytoliths of conifers and palms (Prasad et al. 2005). The grass phytoliths pushed the origin of the Poaceae family back into the age of the dinosaurs.
Benton's conclusion
Dinosaurs did eat angiosperms, but the timing mismatch in Figure 5 argues against the idea that dinosaur herbivory drove the angiosperm revolution, or vice versa. Angiosperms succeeded mainly on their own merits: double fertilization, coevolution with insect pollinators, and above all the Late Cretaceous jump in stomatal and vein density, which gave them an edge in photosynthetic efficiency over the gymnosperms they eventually replaced.
Source: Benton, M. J. (2024). The dinosaur boom in the Cretaceous. Geological Society, London, Special Publications, 544, 453-475. doi.org/10.1144/SP544-2023-70. Open access under CC BY 4.0. Figures 1, 4a (cropped from Figure 4), and 5 reproduced under the terms of the license.
The fern spike: signature of catastrophe
When everything died, ferns were the first to return. The "fern spike" is the clearest palynological signal of ecological collapse in the fossil record.
First identified by Tschudy et al. (1984) in rocks from the Hell Creek Formation (Montana, USA), the "fern spike" consists of an abrupt increase in fern spores in the palynological record: from about 25% to over 70 to 100% of palynomorphs, just above the iridium layer deposited by the Chicxulub impact. Vajda and Bercovici (2014) confirmed the pattern on a global scale, documenting equivalent fern spikes in New Zealand and Japan.
The K-Pg fern spike lasted between 1,000 and 10,000 years, an extremely brief geological interval that reveals the scale of the devastation. Analogous phenomena were recorded after the end-Permian extinction (~252 Ma), with lycophytes dominating the record, and at the end of the Triassic (~201 Ma). Full recovery of diversified forests took between 100,000 and 1,000,000 years.
Why do ferns dominate after catastrophes?
Microscopic spores: produced by the millions, wind-dispersed, no dependence on pollinators
Tolerate low light: survive the "impact winter" caused by atmospheric dust
Rapid colonization: grow in poor, acidic, and newly exposed soils
No animal dependence: reproduction completely independent of fauna
Post-impact recovery sequence
Vegetation collapse, iridium layer
Fern spike: ferns dominate the landscape
Gradual return of angiosperms and conifers
Full recovery of diversified forests
Modern analogues: after the eruption of Krakatoa (1883) and Mount St. Helens (1980), ferns were among the first plants to establish themselves on volcanic ash.
And in this world of conifers, flowers, and ferns, one family of plants was quietly beginning its rise...
The rise of grass
From a marginal plant in the Cretaceous to the dominant biome in the Miocene: the trajectory of grasses over 100 million years.
Origin of grasses
Molecular estimates place the origin of the Poaceae family between 80 and 100 million years ago, still in the Cretaceous period. The oldest grass fossil was found in Myanmar amber (~100 Ma). The first grasses were small understory plants, growing in the shade of conifers, ferns, and angiosperms that dominated the Mesozoic landscape.
Dinosaurs ate grass
In 2005, Prasad and colleagues published a discovery in Science that shook paleontology: phytoliths from at least five different types of grasses were found in titanosaur coprolites from the Lameta Formation, central India. Giant sauropods of the Late Cretaceous were already including grasses in their diet. Grasses were not dominant, but they were part of the available vegetation.
Grasses survive the extinction
The Chicxulub impact killed the non-avian dinosaurs, but grasses made it through the catastrophe. As herbaceous plants with basal meristems (growth points close to the ground), they were more resistant to destruction than trees. This same trait makes grasses resistant to fire, grazing, and drought.
Silent diversification
In the warm, humid world of the Paleocene and Eocene, forests dominated. Grasses remained minor components of the vegetation for tens of millions of years, slowly diversifying in understories and along riverbanks. The Bambusoideae and Ehrhartoideae subfamilies (which include rice) differentiated during this period. The phytolith record shows a gradual increase in grass presence.
First open grasslands
Global cooling after the Antarctic glaciation (~33.9 Ma) and falling CO₂ levels created drier, more seasonal conditions. Forests retreated. For the first time, open habitats dominated by grasses began to appear in parts of South America and Africa. The phytolith record shows the transition from forest vegetation to mixed.
The Grass Revolution
The great moment. Global cooling, the drop in atmospheric CO₂ below ~500-800 ppm, and increased seasonality created ideal conditions for the explosion of C4 grasses. Open grasslands replaced forests across vast continental areas of Africa, the Americas, and Asia. Fauna was transformed: horses evolved from three-toed forest animals to single-hoofed grazers with hypsodont teeth (high-crowned, resistant to silica wear from grasses). Ruminants diversified. Swift predators thrived on the open plains. It was the greatest restructuring of terrestrial ecosystems since the extinction of the dinosaurs.
The dominant biome
Grasses cover approximately 40% of the Earth's land surface (excluding Greenland and Antarctica). The Poaceae family has about 12,000 species. Wheat, rice, corn, sorghum, sugarcane, and rye are all grasses: they sustain the caloric foundation of human civilization. Natural grasslands are now considered the most threatened biome on the planet, with over 70% already converted to agriculture.
Did dinosaurs eat grass?
For decades, paleontology assumed that grasses only appeared after the dinosaurs. In 2005, titanosaur coprolites in India proved otherwise.
What are phytoliths?
Phytoliths are microscopic silica (SiO₂) structures that form inside plant cells. Each grass subfamily produces phytoliths with distinct diagnostic shapes: bilobate, saddle-shaped, cross-shaped. When the plant dies or is digested, phytoliths resist decomposition and remain in the soil, sediments, or coprolites for millions of years. They are like botanical "fingerprints" that survive geological time.
What they found
In the coprolites from the Lameta Formation (central India, ~67 Ma), Prasad et al. identified phytoliths from at least five distinct grass lineages, including representatives of the Bambusoideae and Ehrhartoideae subfamilies (the rice subfamily). The diversity found indicates that the family was already well differentiated before the extinction of the dinosaurs. In 2011, a second study by the same group, published in Nature Communications, demonstrated that the rice tribe (Oryzeae) had already diversified in the Late Cretaceous.
Before vs. after the discovery
Old view
"Grasses arose in the Cenozoic, after the extinction of the dinosaurs. No dinosaur ever ate grass. Open grasslands are a post-dinosaur phenomenon."
Current view
"Grasses arose in the Cretaceous (80-100 Ma) and coexisted with dinosaurs for at least 30 million years. Titanosaurs ate grass. Open grasslands came later, in the Miocene."
C4 Photosynthesis: the secret weapon
Most plants use the C3 photosynthetic pathway. Some grasses developed something better.
What is the C4 pathway?
A modified photosynthetic mechanism that concentrates CO₂ around the RuBisCO enzyme, reducing photorespiration. It is more efficient than the C3 pathway under conditions of intense heat, high light, drought, and low atmospheric CO₂.
Extreme convergent evolution
C4 photosynthesis evolved independently at least 22 to 24 times within grasses, one of the most remarkable examples of convergent evolution in plants. The selective pressure from falling CO₂ in the Oligocene-Miocene favored this mechanism in multiple lineages simultaneously.
Disproportionate impact
C4 grasses represent only ~3% of plant species but account for 23-25% of global terrestrial primary productivity. They dominate tropical and subtropical savannas. Corn, sugarcane, sorghum, and millet are all C4.
How grasses changed the Earth
A plant family that reshaped ecosystems, soils, fire regimes, and the evolution of animals.
Grass-fire cycle
Grasses produce dry, continuous, and flammable biomass that burns easily and regenerates rapidly from underground meristems. Fire kills young trees, but grasses resprout in weeks. This creates a feedback loop: more grasses generate more fire, which prevents forest encroachment, which favors more grasses. The expansion of grasslands in the Miocene coincides with increased charcoal deposits in the sedimentary record.
The most fertile soils on the planet
Grasses possess dense, fibrous root systems that penetrate up to 2-3 meters deep. These roots contribute massive amounts of organic carbon to the soil, creating the world's most fertile soils: Mollisols (Chernozems). The North American Great Plains, the Ukrainian steppe, and the Argentine Pampas owe their fertility to millions of years of organic matter accumulation by grasses.
Faunal transformation
The Miocene Grass Revolution restructured fauna on every continent. Horses evolved from Mesohippus (3 toes, low teeth for leaves) to Equus (single hoof, hypsodont teeth for abrasive silica-rich grasses). Ruminants such as bovids and cervids diversified with multi-chambered stomachs to digest cellulose. Cursorial predators (runners) like felids and canids thrived on the open plains.
Grasses by the numbers
~40%
of the land surface
12,000
known species
100 Ma
oldest fossil
22-24x
times C4 evolved independently
5+
grass types in titanosaur coprolites
>70%
of native grasslands already lost
References
Grasses
Prasad, V., Strömberg, C.A.E., Alimohammadian, H. & Sahni, A. (2005). Dinosaur coprolites and the early evolution of grasses and grazers. Science, 310(5751), 1177-1180.
Strömberg, C.A.E. (2011). Evolution of grasses and grassland ecosystems. Annual Review of Earth and Planetary Sciences, 39, 517-544.
Cerling, T.E. et al. (1997). Global vegetation change through the Miocene/Pliocene boundary. Nature, 389(6647), 153-158.
Prasad, V. et al. (2011). Late Cretaceous origin of the rice tribe provides evidence for early diversification in Poaceae. Nature Communications, 2: 480.
Mesozoic forests and dinosaur diet
Hummel, J. et al. (2008). In vitro digestibility of fern and gymnosperm foliage: implications for sauropod feeding ecology. Proc. R. Soc. B, 275(1638), 1015-1021.
Bomfleur, B., McLoughlin, S. & Vajda, V. (2014). Fossilized nuclei and chromosomes reveal 180 million years of genomic stasis in Royal Ferns. Science, 343(6177), 1376-1377.
Bennettitales
Crane, P.R. (1985). Phylogenetic analysis of seed plants and the origin of angiosperms. Annals of the Missouri Botanical Garden, 72(4): 716-793.
Crepet, W.L. (1974). Investigations of North American cycadeoids: the reproductive biology of Cycadeoidea. Palaeontographica Abt. B, 148: 144-169.
Angiosperm revolution
Gomez, B. et al. (2015). Montsechia, an ancient aquatic angiosperm. PNAS, 112(35), 10985-10988.
Sun, G. et al. (2002). Archaefructaceae, a new basal angiosperm family. Science, 296(5569), 899-904.
Flower-pollinator coevolution
Poinar, G.O. & Danforth, B.N. (2006). A fossil bee from Early Cretaceous Burmese amber. Science, 314(5799), 614.
Peñalver, E. et al. (2012). Thrips pollination of Mesozoic gymnosperms. PNAS, 109(22), 8623-8628.
Fern spike
Tschudy, R.H. et al. (1984). Disruption of the terrestrial plant ecosystem at the Cretaceous-Tertiary boundary. Science, 225(4666), 1030-1032.
Vajda, V. & Bercovici, A. (2014). The global vegetation pattern across the Cretaceous-Paleogene mass extinction interval. Global and Planetary Change, 122, 29-49.